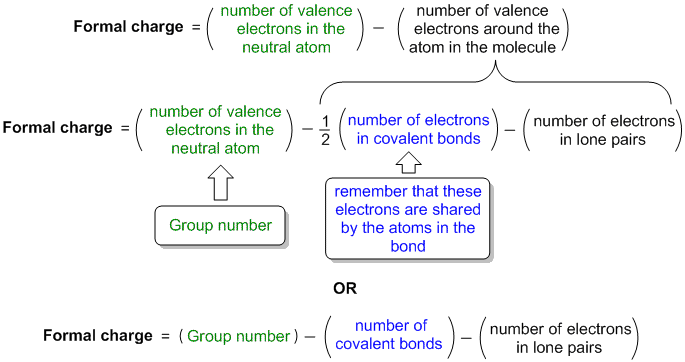
The structure is surrounded by brackets and has a superscripted negative sign. The right structure shows a carbon atom with two lone electron pairs double bonded to a sulfur atom that is double bonded to a nitrogen atom with two lone electron pairs. Below this structure are the numbers negative two, positive one, and zero. 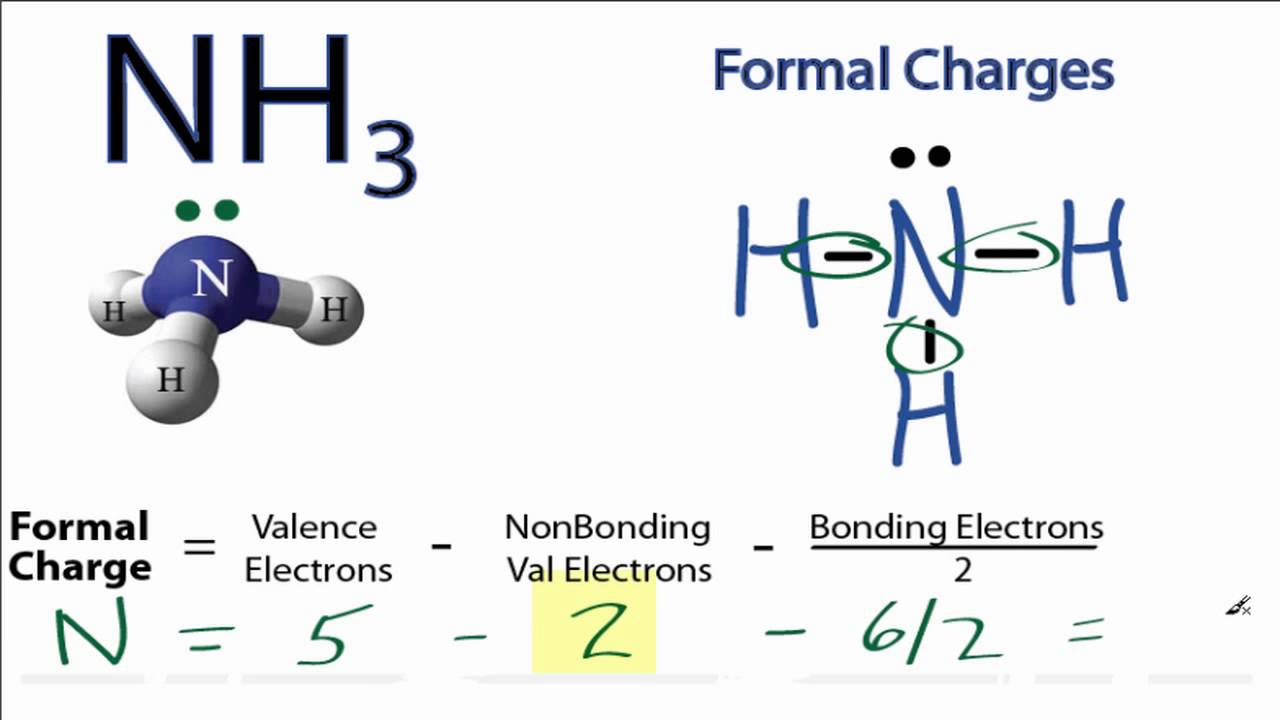
The structure is surrounded by brackets and has a superscripted negative sign. We can double-check formal charge calculations by determining the sum of the formal charges for the whole structure. The middle structure shows a carbon atom with two lone pairs of electrons double bonded to a nitrogen atom that is double bonded to a sulfur atom with two lone electron pairs. Formal charges should be as small as possible (comparing the absolute value. Below this structure are the numbers negative one, zero, and zero. The sum of the formal charges must equal to the total charge on the molecule or ion. The structure is surrounded by brackets and has a superscripted negative sign. So, for as long as you have a complete Lewis structure and periodic table handy, you can quickly find the formal charge of any atom in a molecule. For example, the right oxygen in the (correct) NO 2 structure (see Figure 4) has a -1 charge, so it can either attract positively charged atoms/molecules and/or donate electrons. While its easy to just say that, its important to place. This helps us understand/predict the kind of reactivity the molecule will have. The formal chemical charge of Ammonia (NH3) is zero, it doesnt actually have a chemical charge. The top row is labeled, “Structure” and depicts three Lewis structures and the bottom row is labeled, “Formal charge.” The left structure shows a carbon atom double bonded to a nitrogen atom with two lone electron pairs on one side and double bonded to a sulfur atom with two lone electron pairs on the other. By calculating the formal charge, we can determine where (if any) charges are within the molecule. Boost your Chemistry grade with Calculating Formal. Get instant feedback, extra help and step-by-step explanations. Two rows of structures and numbers are shown. Practice Calculating Formal Charge with practice problems and explanations. Possible Lewis structures and the formal charges for each of the three possible structures for the thiocyanate ion are shown here: 
In order to use the formula charge formula, we must observe the Lewis Dot Structure for. The formal charge for the Carbon atom, attached with the Oxygen atom. The oxidation number of each atom can be calculated by subtracting the sum of lone pairs and electrons it gains from bonds from the number of valence electrons. Step 1: Draw the Lewis Dot Structure for the compound given in order to calculate the formal charge. The formal charges present in each of these molecular structures can help us pick the most likely arrangement of atoms. The formal charge of the Carbon atom, attached with Hydrogen atoms (4 0 (1/2×8)) 0.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
